
In Section 1.3 'Elements and the Periodic Table', we introduced the periodic table as a tool. 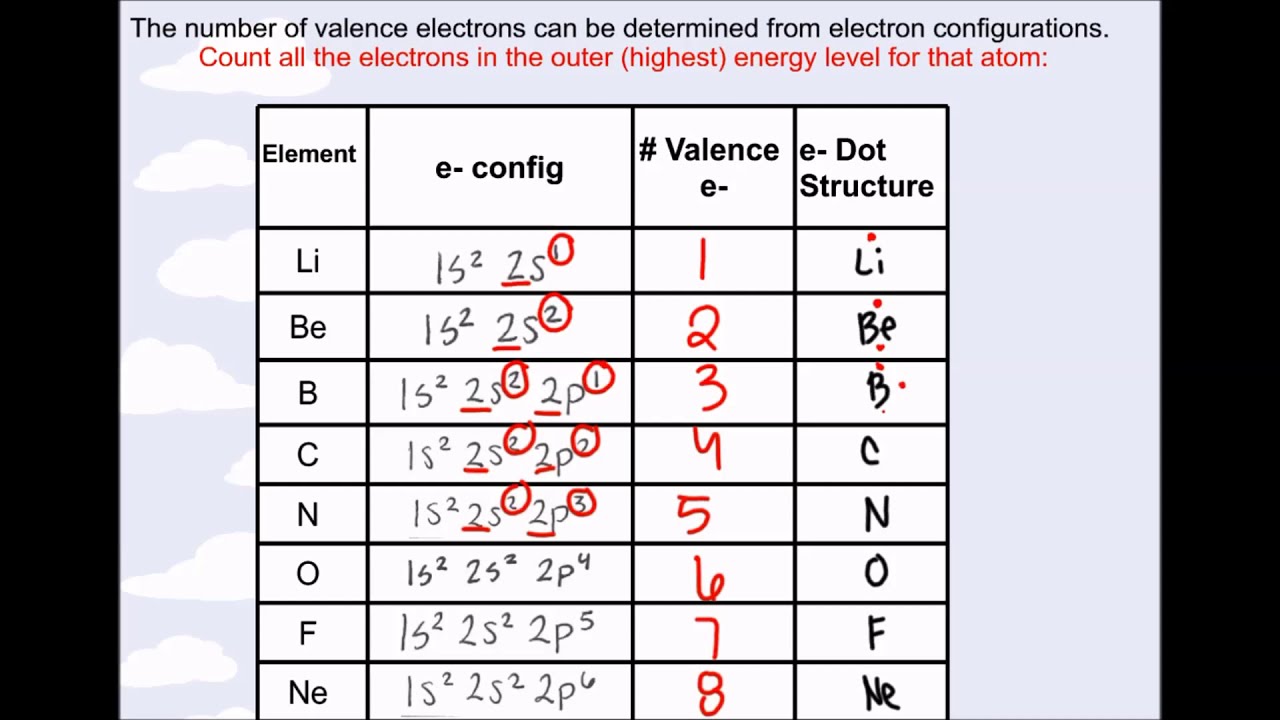
By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistryand they do. The elements in group-18 of the periodic table are inert gases. The electron configuration of an element is the arrangement of its electrons in its atomic orbitals. They all have a similar electron configuration in their valence shells: a single s electron. Therefore, the valence electrons of xenon are eight. This electron configuration shows that the last shell of xenon has eight electrons. Covalent radius mostly decreases as we move left to right across a period because the effective nuclear charge experienced by the electrons increases. The ground-state electron configuration of xenon is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6. This means that the first two electrons occupy the K shell, the next eight electrons fill the L shell, the following 18 electrons occupy the M shell, and the remaining 15 electrons fill the N shell. B Thus beryllium has an He s2 electron configuration. Ruthenium, with its atomic number 44, has an electron configuration of 2 8 18 15 1. Although hydrogen is not an alkali metal, its 1 s1. Hydrogen and helium are placed somewhat arbitrarily. Because each orbital can have a maximum of 2 electrons, there are 2 columns in the s block, 6 columns in the p block, 10 columns in the d block, and 14 columns in the f block. The electron configuration for the first 10 elements. The electron configurations of the elements are in Figure 6.9.2. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n 2. Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. Covalent radius increases as we move down a group because the n level (orbital size) increases. Solution: A The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. Five 3 d orbitals are filled by the next 10 elements, the transition metals, followed by three 4 p orbitals. Electron configurations allow us to understand many periodic trends. Consequently, the electron configuration of potassium, which begins the fourth period, is 4 s 1, and the configuration of calcium is 4 s 2. Mathematically, configurations are described by Slater determinants or configuration state functions. Accordingly, the 4 s orbital is filled prior to the 3 d orbital because of shielding and penetration effects. Electronic configurations describe each electron as moving independently in an orbital, in an average field created by all other orbitals. The order in which the orbitals are filled is indicated by the diagonal lines running from the upper right to the lower left. For example, the electron configuration of lithium, 1s2s, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. Subshells corresponding to each value of n are written from left to right on successive horizontal lines, where each row represents a row in the periodic table. Electron configuration notation provides us with information about the basic energy levels and sublevels that electrons occupy. Electron configurations describe where electrons are located around the nucleus of an atom. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Electron configuration chart of all Elements is mentioned in the table below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
